Background & Context
About Boswellia serrata
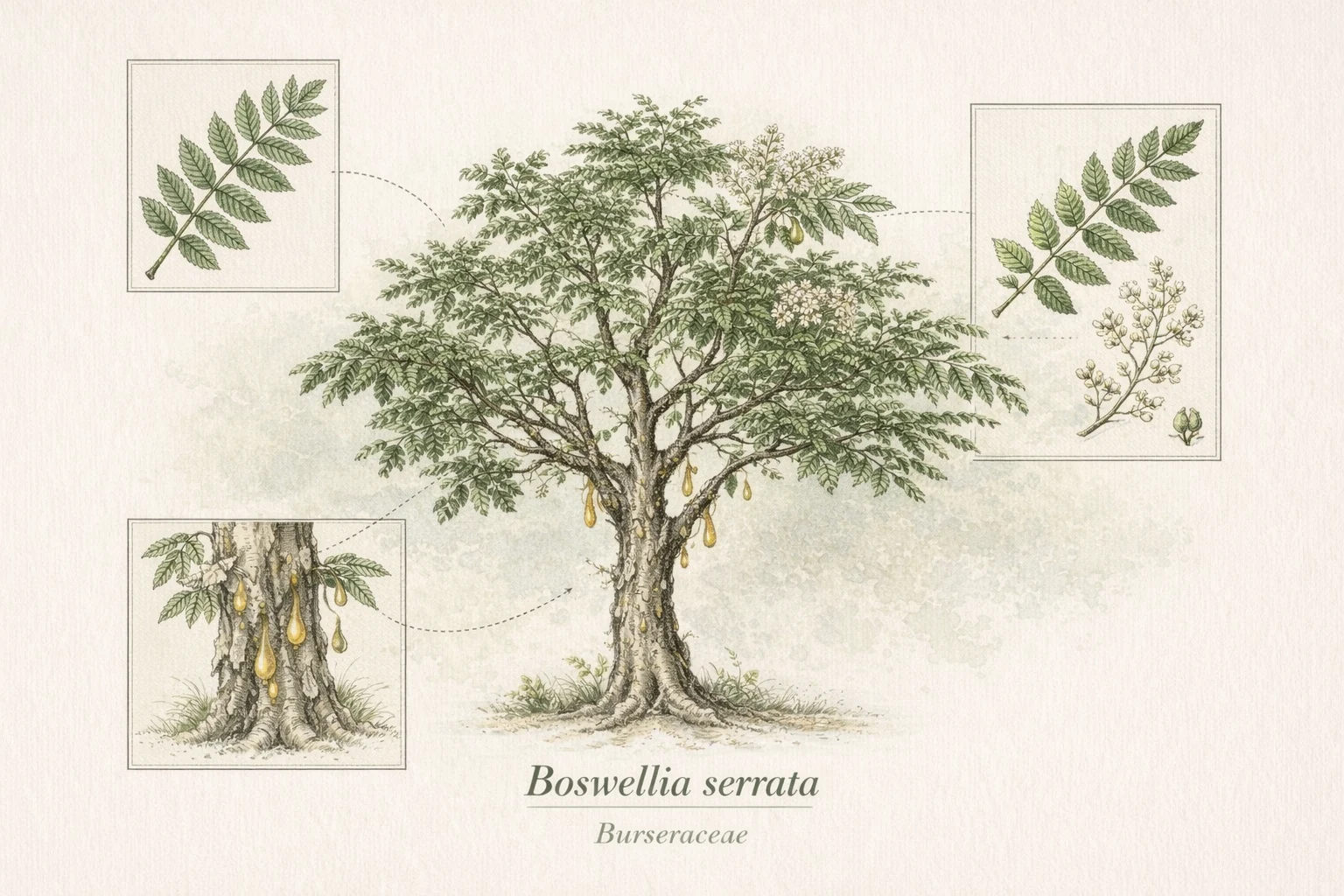
Boswellia serrata is a deciduous tree native to the dry hill forests of India, Pakistan, and Bangladesh. The oleogum resin extracted from incisions in its bark — commonly referred to as frankincense or Indian olibanum — has been used in Ayurvedic medicine for centuries, documented in classical texts including the Sushruta Samhita under the name Shallaki.
Over 20 Boswellia species have been identified. Each species produces a distinct chemical profile; research findings from one species do not necessarily apply to others. The majority of published clinical research has investigated Boswellia serrata specifically.
Read the Historical Record
20+
Boswellia species identified; B. serrata is the most studied in clinical trials
Centuries
of documented traditional use in Ayurveda; precise historical dating is uncertain
30–65%
Boswellic acid content in standardized research extracts; unstandardized products vary widely
AKBA
Most studied boswellic acid; inhibits 5-LOX in vitro; clinical significance still under investigation